 
The atomic number represents the number of protons in the nucleus In nuclide notation, the bottom-left number is the atomic number. How many neutrons are there in atoms of oxygen-18?.How many protons are there in atoms of oxygen-18?.The isotope oxygen-18 has the nuclide symbol 1 8 8 O. Using our understanding of atomic number and mass number, we can represent an atom of boron that contains five protons, six neutrons,Īnd five electrons with the following nuclide notation.Įxample 2: Deducing the Number of Protons and Neutrons and the Mass Number from Nuclide Notation n u m b e r o f n e u t r o n s n u m b e r o f n e u t r o n sĪn atom of calcium has 22 neutrons in its nucleus. Number given in the question into the mass number equation and solve for the number of neutrons:Ĥ 2 = 2 0 + 2 2 =. We can substitute the number of protons and the mass We have already established that an atom of calcium contains 20 protons. M a s s n u m b e r n u m b e r o f p r o t o n s n u m b e r o f n e u t r o n s = +. The following equation can be used to calculate the mass number: The mass number is the total number of nucleons in the nucleus of an atom, isotope, or ion. Therefore,Īn atom of calcium with an atomic number of 20 has 20 protons in its nucleus. The atomic number represents the number of protons in the nucleus of all atoms, isotopes, and ions of the element. How many neutrons are there in its nucleus? Answer I mage Credits: Wikimedia Commons/The Sun.Example 1: Converting from the Mass Number and the Atomic Number to the Number of NeutronsĪn atom of calcium has a mass number of 42 and an atomic number of 20. Defining and calculating atomic mass is not difficult you just need the correct tools and an understanding of the chemistry terms. The atomic mass of the compound H2O is 18.0098.Ītomic mass is a basic chemistry concept. There is only one oxygen atom: 15.9994.Ĥ. Example: You have two hydrogen atoms times the mass. Next multiply the number of atoms of each element by the atomic mass of the element. Next, check the periodic table for the atomic mass of the elements used. Write out the chemical formula of the compound. To calculate the atomic mass of a compound you will need a calculator, periodic table, pencil and paper.ġ. 
The mass number shown on the periodic table is an average of the mass of the isotopes of the element taking into account the abundance of the isotope’s occurrence in nature. Isotopes are forms of an element with the same atomic number (same number of protons) but different atomic masses due to different numbers of neutrons. The reason for this is that most elements consist of two or more isotopes. For example, hydrogen has an atomic mass of 1.00794. You may wonder why these numbers are usually not whole numbers. These are used to calculate the atomic mass of compounds. The average atomic masses of all known elements are listed on the periodic table. Using such small numbers in calculations would be too complicated. Grams are the standard unit to express mass but because atoms are so small, grams or milligrams are not convenient to express the numbers of atomic mass. The atomic mass of hydrogen is 1 amu an atom of hydrogen is 1/12 the mass of an atom of C-12. The atomic mass of an atom is the number of times it is heavier than 1/12 an atom of C-12. An atomic mass unit is equal to 1/12 the mass of a carbon -12 atom. Scientists refer to the mass of atoms in atomic mass units or amu. However, mass is the amount of matter in an object and weight is the amount of mass times the gravitational pull on an object which makes atomic mass the preferred term. Atomic mass numbers for all known elements are listed on the periodic table.Ītomic mass and atomic weight are terms used interchangeably. The electrons outside the nucleus can be ignored as their mass is so tiny as to be negligible. 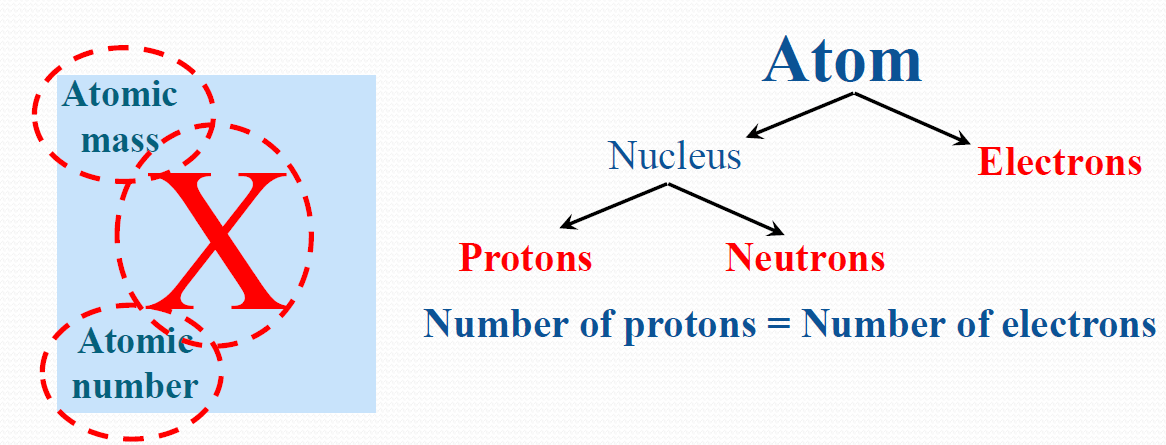
The definition of atomic mass is the approximate sum of protons and neutrons in an atom’s nucleus. “How do I calculate atomic mass?” is a very common question for beginning chemistry students. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
